 
And its linear molecular geometry, as per the principle of VSEPR. It fits the molecular geometry of AX2 since carbon is bonded to two atoms. HCN has the geometry of AX2 molecule, where the central atom is A, and the number of atoms bound to the central atom is X. Prevents oxygen from using the body cells owing to which the cells die.Įvery molecule’s molecular geometry helps to explain its dimensional structure and its form.In open fields, cyanide gas evaporates instantly, making it less dangerous outdoors.Especially, in confined areas, it is risky.Cyanide gas breathing does the most pain, but it may also be poisonous to swallow it.The degree of cyanide toxicity depends on the quantity and duration of cyanide to which an individual is exposed.In its exterior valence shell, hydrogen has two electrons, and the other two electrons are non-bonding electrons. Similarly, nitrogen has a full octet and it only requires three electrons to complete the octet it gets by exchanging the electrons with carbon. Now, by forming a single hydrogen bond and a triple nitrogen bond, Carbon has a full octet. Thus, carbon will have four electrons, hydrogen will have one, and nitrogen will have five electrons inside the atom. If the atoms have been arranged, start putting the dots around individual atoms representing the valence electrons. Then, you need to position the atoms of hydrogen and nitrogen on both ends of the carbon atom. Carbon takes the middle position as it is the least electronegative. It also helps to consider the bonds formed in the molecule and the electrons that do not take part in the forming of either bond.įirstly to make the dot structure you need to decide the center atom and then position the leftover atoms in the structure of HCN. The Lewis HCN structure helps to explain the configuration in the molecule of valence electrons around the atoms. 
Products of combustion are compounds that are given off as objects ignite.
0 Comments
Leave a Reply. |
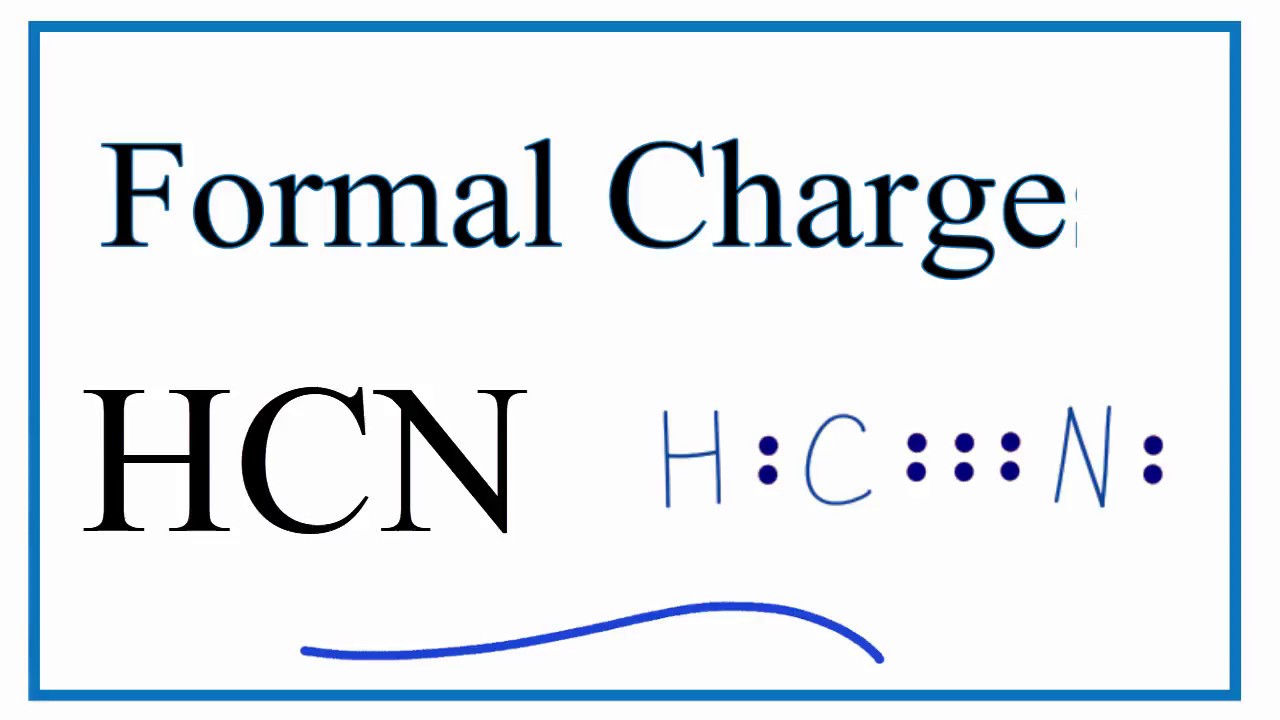
 RSS Feed
RSS Feed
